Advancing the Future of Oncology
Our mission is to transform cancer treatment by delivering safe, effective, and affordable therapies, making life-saving innovation accessible to patients worldwide.
Our vision is to lead the next generation of CAR-T therapies, setting new benchmarks in safety, efficacy, and cost-effectiveness. Through cutting-edge science, precision engineering, and a commitment to accessibility, we aim to bring curative immunotherapies to patients globally and redefine the future of oncology.
Follow us to stay up to date — click below:
Amatista Biotech: Innovation in CAR-T Biotechnology
CAR-T (Chimeric Antigen Receptor T-cell) therapy is revolutionising cancer treatment. It works by reprogramming a patient’s own immune cells to recognise, target, and destroy cancer with unmatched precision.
T cells are collected from the patient, genetically engineered in the lab to specifically recognise cancer cells, and then reinfused to mount a targeted and potent immune response.
As a tailored, gene-engineered therapy, CAR-T represents the forefront of personalised medicine, adapting to each patient’s tumour profile with unprecedented accuracy.
Traditional CAR-T therapies can trigger severe side effects such as cytokine release syndrome and neurotoxicity. These reactions arise when engineered T cells become overactivated in the body. Such uncontrolled immune responses can limit treatment safety and applicability.
✓
Positive preclinical data
✓
Patented technology
✓
Collaborations with 3 top-tier institutions in Europe and the USA

Our Innovation: The Universal Safety Switch
Amatista Biotech’s technology relies on a universal CAR-T kinase switch, a next-generation safety mechanism that is reversible and dose-dependent, allowing clinicians to control toxicity without destroying the therapeutic cells. This innovation makes CAR-T therapies safer, more flexible, and more accessible to patients worldwide.
Incorporating a molecular “switch” allows clinicians to modulate CAR-T cell activity in real time. The switch can temporarily pause, reduce, or fully shut down the cells if toxicity emerges.
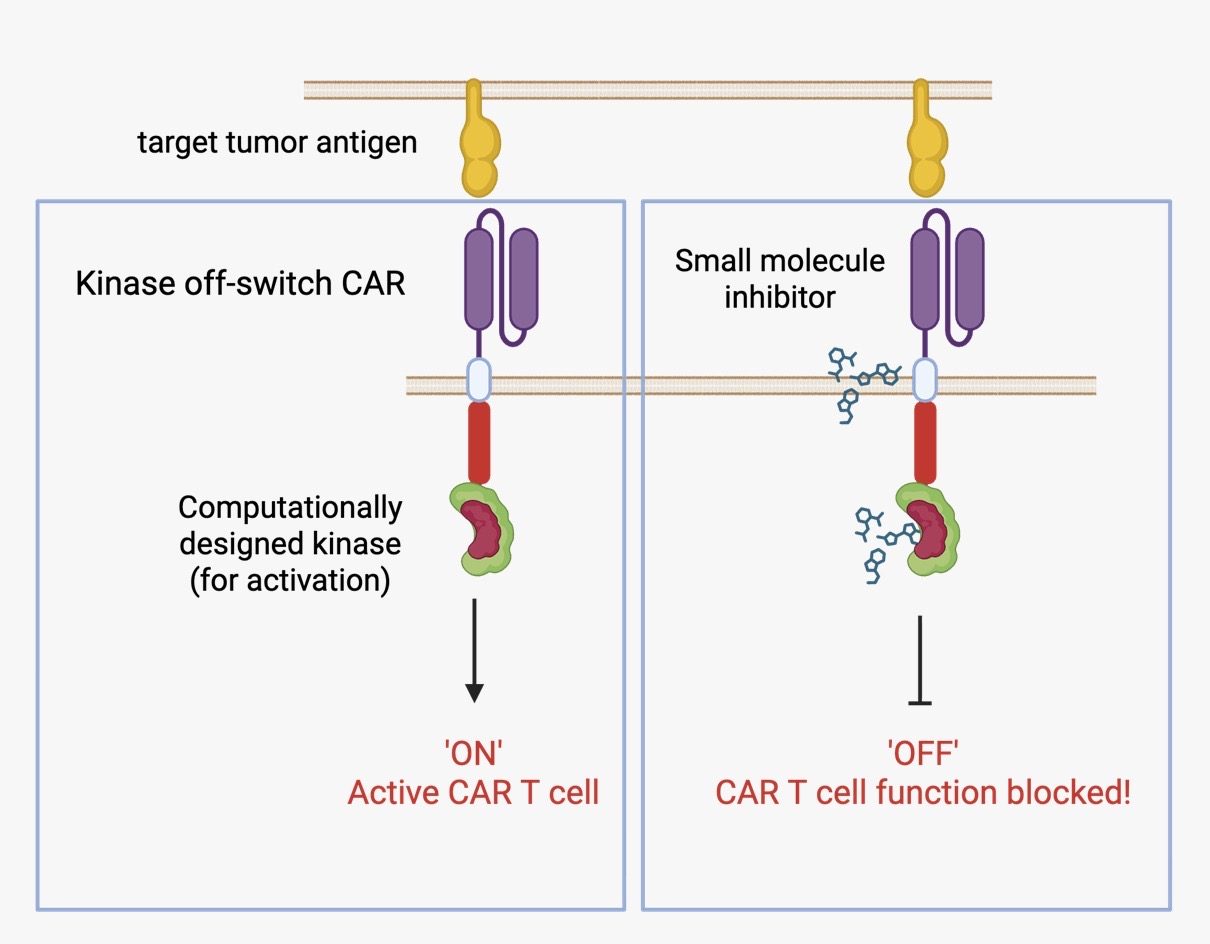
Mechanism of Action
Our switch platform involves knocking out an endogenous tyrosine kinase critical for T-cell activation and replacing it with a computationally modified variant sensitive to erlotinib. In the absence of the drug, the CAR-T cells function normally (‘ON’ state). When erlotinib is administered, the engineered kinase is inhibited, temporarily halting T-cell activation/function (‘OFF’ state). This rapid, robust and reversible control mechanism allows clinicians to pause CAR-T function in the event of toxicity.

Our Strategy
Our universal switch is designed to complement, not compete with existing and developing CAR-T therapies. It serves as a versatile platform that companies can integrate to enhance the safety, control, and efficacy of their treatments, accelerating the next generation of CAR-T innovations.
We are also advancing CAR-T therapies against PSMA, a well-validated antigen in prostate cancer. With no approved CAR-T treatments currently available for this indication, Amatista Biotech is positioned as a first-mover, addressing a significant unmet medical need in solid tumours.
Get in Touch
Reach out to us via email at info@amatistabiotech.com or using the contact form below.

